EU Greenlights Moderna’s mNEXSPIKE: Same Vaccine Category, Smaller mRNA Payload, Lower Dose
This isn’t a delivery revolution—it’s a pragmatic industrial optimization: truncate the antigen, cut the dose, and buy back capacity, tolerability, and commercial viability.
The Signal
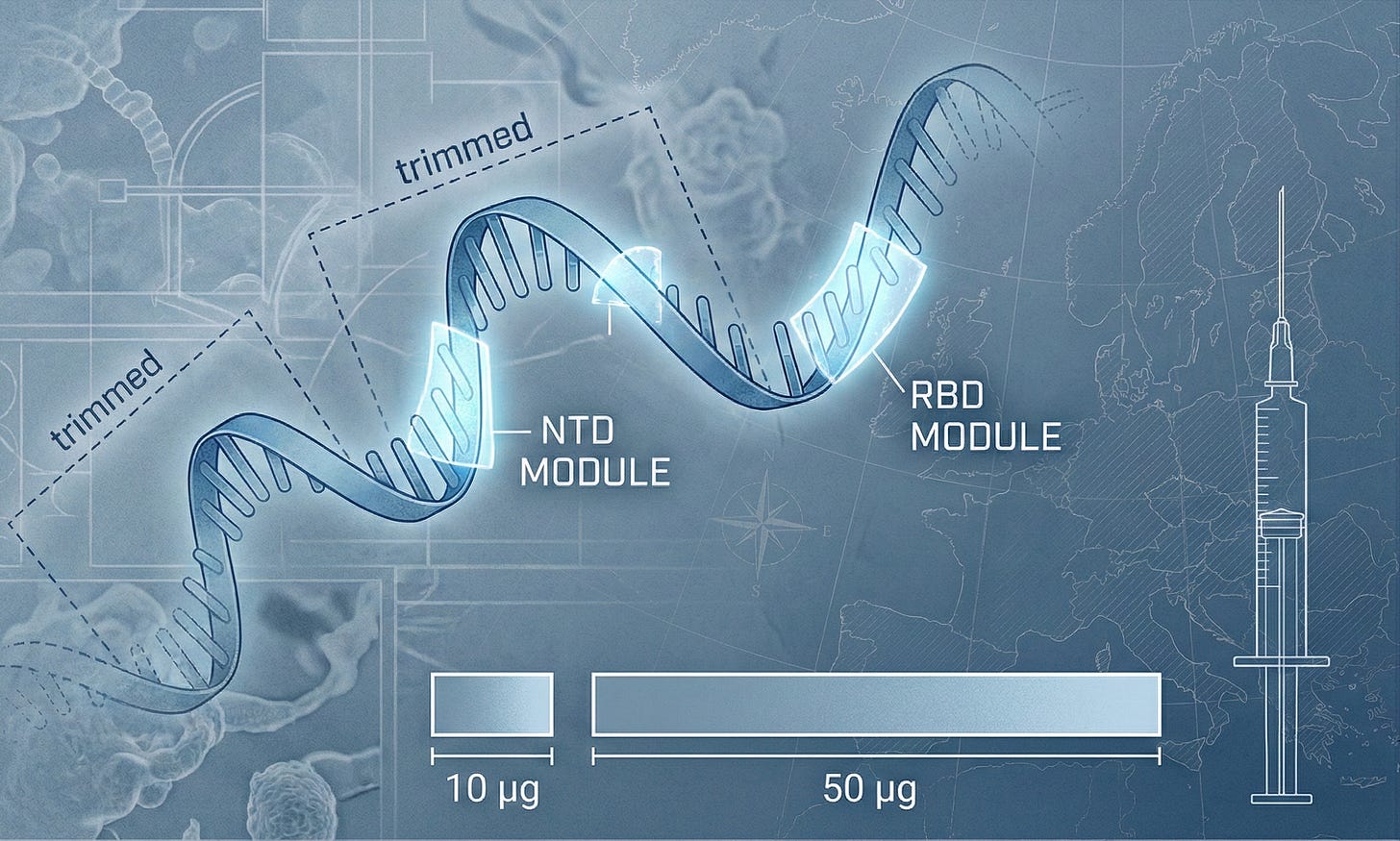
Feb 17, 2026 — Moderna announced that the European Commission (EC) granted marketing authorization in the EU for mNEXSPIKE (mRNA-1283), its next-generation COVID-19 mRNA vaccine, for active immunization in individuals 12 years and older. The decision was supported by a Phase 3 study (~11,400 participants) comparing mNEXSPIKE 10 μg versus Spikevax 50 μg, using a non-inferiority framework. Moderna disclosed 9.3% higher relative vaccine efficacy in the overall population and 13.5% higher in adults ≥65 years (descriptive analysis), alongside fewer local reactions.
RapidGene Take
The real signal isn’t how “new” mNEXSPIKE is—it’s how ruthlessly practical it is. The industry has moved past “platform solves everything” hype. What wins now is COGS, capacity, and willingness-to-get-jabbed. A 10 μg vs 50 μg comparison is basically a message to the supply chain: the same factories can output a lot more doses, and to vaccinators: reactogenicity (at least local) can be pushed down without compromising immunogenicity claims. Don’t romanticize it, though: this is product engineering, not a miracle. EU approval is a ticket to the arena, not proof of demand. The real fight is procurement, reimbursement, and how fast “seasonal COVID” purchasing patterns normalize.
Expanded Read
Technically, mNEXSPIKE’s differentiator is clear: it does not encode full-length Spike. EMA’s public product information describes mNEXSPIKE’s mRNA as encoding a membrane-anchored, linked N-terminal domain (NTD) and receptor-binding domain (RBD). In plain terms, this is “subtractive design”: keep the neutralization-relevant regions, drop the rest of the payload.
That “truncated antigen” choice matters more for industrial reality than for flashy science:
Shorter mRNA (inference): Encoding only NTD+RBD implies a shorter transcript, which can reduce burden across IVT and downstream processing (reagent usage, chromatography load, filtration throughput). Even if you distrust the “shorter is easier” narrative, the 10 μg dose is a hard, multiplicative capacity lever—full stop.
Dose economics: mRNA vaccine costs aren’t only the RNA. The bill is the whole DP chain—lipids, buffers, aseptic fill/finish, cold chain, and QC release. But lower dose can cascade into real operational flexibility: lower API demand, potentially wider process windows, and more room for batch-to-batch consistency strategies.
Tolerability isn’t “nice to have”—it’s the sellability threshold: Moderna reported similar systemic reactions versus Spikevax, with fewer local reactions. You can stay cynical about PR language, but commercially the enemy is not antibody titers anymore—it’s people opting out of another shot.
Regulatorily, the EU pathway is textbook: CHMP issued a positive opinion (Moderna references December 2025 timing), followed by EC marketing authorization. EMA’s framing is restrained—mNEXSPIKE’s performance is described as comparable to Spikevax, supporting a “not worse” story rather than a “breakthrough” claim. That’s how iterative products get through: don’t expect a new narrative, deliver clean comparator data.
Competitively, this is also a tell: while the industry chases the next big mRNA story (tumor vaccines, extrahepatic delivery, targeted LNPs), Moderna is reinforcing its edge in the most cash-flow-relevant arena—respiratory vaccines—by doing what mature modality leaders do: optimize the product for manufacturing and uptake. But again, don’t confuse authorization with victory: procurement dynamics, payer behavior, and how combo-respiratory products evolve in a more regulatorily sensitive environment will determine the real outcome.
Keywords
mNEXSPIKE; mRNA-1283; EU marketing authorization
Disclaimer
RapidGene Insights provides technical and strategic opinions based on public data. This is not investment advice. If you believe our comments lacks context, please contact us.